Any student having prepared for matriculation board exams will expect a stream to opt for in the senior secondary education level. If he happens to take the science stream, the subjects he will need to be good at are obviously physics, chemistry and maths. As this article focuses on SSC chemistry syllabus, we will explore chemistry topics in a brief overview.
Chemistry is a remarkable subject, perhaps much more than others, to study. It constitutes the basis of everything by which we are surrounded, including ourselves, for everything is made up of atoms and molecules.
Chemistry includes the study of all the materials (from the smallest to the largest) of which the ‘matter’ is made up of. The matter is anything that takes space and has a definite mass.
However, chemistry classes are not limited to studying atoms and molecules. As a subject, it covers an elaborate range of topics. If you are going to prepare for SSC level chemistry, it is important to understand these basic topics.
Keeping your academic needs of chemistry in mind, let’s look at the topics we have selected which you would do well to focus on.
Learn more about some of the amazing chemistry classes in Chennai here.
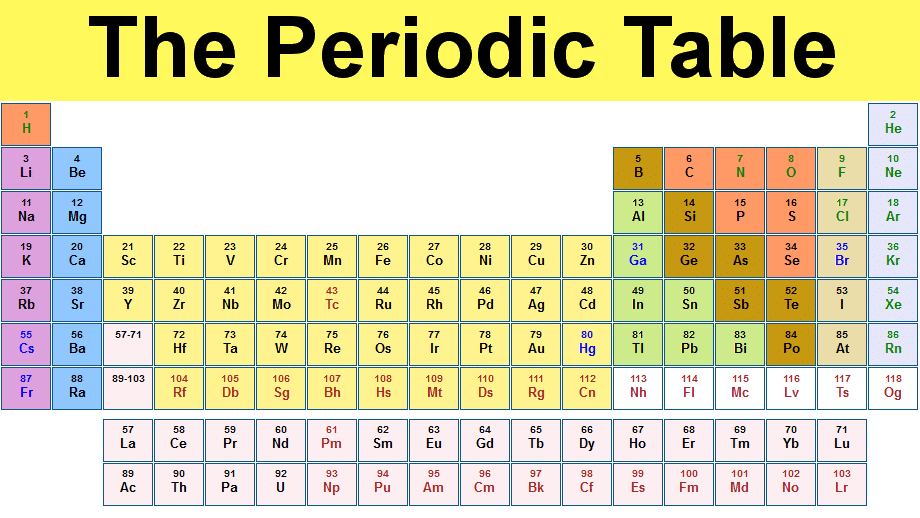

Atomic Structure And The Periodic Table For SSC Level
On the most fundamental level, every chemistry student ought to be good in the knowledge of the atomic structure and the periodic table. In fact, the two go hand in hand as both are needed to understand each other.
On the SSC level, you will be taught about the atom and its structurality; as well as how and by what value the elements are organised on the periodic table.
Atomic Structure
Atoms are the basis of chemistry and everything else in the Universe. The matter is composed of atoms. While by no means the smallest piece of matter, atoms constitute the foundation of basic chemistry.
In the centre of the atom is the positively charged nucleus consisting of protons and neutrons. The three main constituents of an atom are protons, neutrons and electrons. Protons (positively charged) and neutrons (neutral charge) are found in the nucleus of an atom. Electrons (negatively charged) are found in the orbits surrounding the nucleus.
Periodic Table
In 1869, A chemist from Russia Dmitri Mendeleev published the first recognizable periodic table. In chemistry, the periodic table shows the elements arranged in order of atomic number, electron configuration, and recurring chemical properties. Usually arranged in rows, so that elements with the similar atomic structure are put in vertical columns.
Currently, there are 92 natural elements in the periodic table ranging from hydrogen to uranium. Around 26 transuranic elements have been created or observed.
Chemical Changes Or Reactions
Other important SSC chemistry lessons comprise of chemical changes. Chemical reactions are a common phenomenon in chemistry. To properly understand the reasons behind the various reactions, it is important to be familiar with the concept of chemical changes itself.
Definition Of Chemical Changes
A chemical change or reaction is a process where a substance transforms into one or more new and different substances. A chemical change is basically a rearrangement of electrons and atoms.
Unlike physical change, a chemical change cannot be reversed except, in a few cases, through further chemical processes.
In a chemical reaction, there is also a change in the energy of the substance. If in a chemical change heat is released, it is called exothermic reaction, and if the energy (heat) is absorbed, it is known as an endothermic reaction.
It is important to note that in a chemical change, the number of atoms remains the same only their arrangement is altered.
Types Of Chemical Changes
In general, the three most recognized chemical changes are the following:
- Organic chemical change: Organic chemical changes are the one including organic compounds which are primarily composed of hydrogen and carbon. An example would be polymerization.
- Inorganic chemical changes: Chemical reactions in which carbon is generally not included are known as inorganic chemical changes. Mixing acid and base is one example.
- Biochemical changes: Organic chemical changes in living organisms are known as biochemical changes. The reactions are controlled by hormones and enzymes. The most visible example of this type of change is Photosynthesis.

Atmospheric Chemistry
A recent analysis predicting that the temperature of Europe could fall due to the warming of the Arctic is one of the many reports of the emerging climate crisis. The topic of the chemistry of the atmosphere gives handy information about the atmosphere of our planet. Even from a practical point of view, keeping the climate crisis in mind, this topic seems to be all the more important.
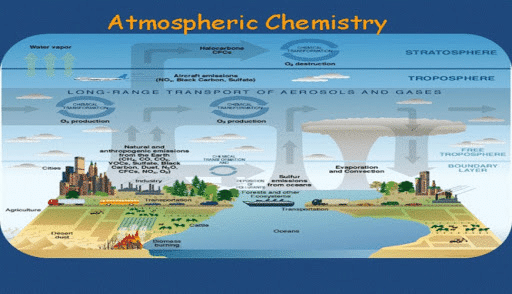
Defining Atmospheric Chemistry
As a branch of atmospheric science, atmospheric chemistry studies the chemical composition of the natural atmosphere of the Earth and other planets. It tells us the way solids, liquids and gases in the atmosphere interact with one another.
According to NASA, Earth’s atmosphere is composed of 78% Nitrogen, 21% Oxygen and 0.4% carbon dioxide. Learning about the atmosphere also helps us in understanding the climate.
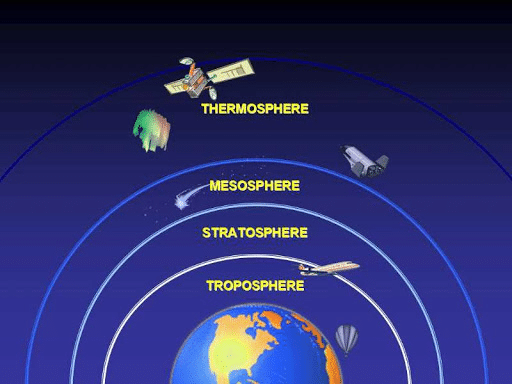
The Four Layers
The division of the atmosphere into four different layers of study are based on temperature variations with the increasing altitude. The four layers of the atmosphere are the troposphere, the stratosphere, the mesosphere and the thermosphere.
Interestingly, in 1995 the Nobel Prize in Chemistry was awarded to the collective works of the atmospheric scientists P. Crutzen, F. S. Rowland and M. Molina.
Click to join a chemistry class here.
Chemical Analysis
While atoms and molecules constitute the basis of the chemistry, its in-depth analysis is called chemical analysis. To be good in chemistry, it is important to cover all the different types of topics in a thorough manner.
The chemical analysis determines the physical properties and chemical composition of a substance.
This topic gives you an idea of various terms often applied in chemistry such as defining pure substances, mixtures, chromatography, spectrometry and so on.
Chemical analysis is divided into two topics: qualitative analysis and quantitative analysis.
Qualitative Chemical Analysis
In qualitative chemical analysis, the objective is to find out or identify the element or a group of various elements comprised within a substance. Qualitative chemical analysis in itself is divided between qualitative organic analysis and qualitative inorganic analysis.
Quantitative Chemical Analysis
In quantitative chemical analysis, the objective is to determine the relative amount or percentage of an element in a sample matter or substance. The methods involved in qualitative chemical analysis are broadly categorised into the chemical and physical analysis.
Becoming an expert in this topic requires a constant need to go through the class notes you already have. Past year examination papers or sample papers can also be of help. Also, it is recommended to prepare separate chemistry notes for SSC from which you can go through various topics in quick time.
Discover other online chemistry classes on Superprof.
Bonding, Structure And The Properties Of Matter
The theories of bonding and structure are used to determine the chemical and physical properties of matter. Bonding relates to how atoms are held together in a given structure. Structure, on the other hand, showcases how atoms can be arranged in different ways.
In chemical bonding, we learn about covalent, ionic and metallic bonding.
- Covalent bonding occurs in nonmetals and their compounds.
- Ionic bonding occurs in the interaction between metals and nonmetals
- Metallic bonding occurs in metals and alloys.
One also gets to learn about the three states of matter - solid, liquid, and gas - and how the transformation from one state to another depends on the bonding and structure of the substance.
The greater the forces between the particles of a substance, the higher the melting and boiling point it will have.
A great deal can be learned about chemistry from this chapter. So those who are looking to prepare for SSC level chemistry should work towards clearing the concepts of this topic.
A Self-help Suggestion
The aforementioned topics are by no means the only topics. However, by and large, they cover some of the most important areas of chemistry. Without these topics, it would be difficult to understand the more complex theories of chemistry. One can also say that these topics form the base of the chemistry of which all the subsequent SSC chemistry syllabus are the superstructure.
You’d do well to revise all these topics, at least, before stepping into the stream of science.
To help yourself, you can also opt for a coaching class or a private tutor to assist you to work on the subject. Coaching centres often conduct mock tests, which will be good to test your skills.
Help From Internet
For extended reading, the internet also can be of great help. You can easily get central board syllabus: for instance, ICSE or CBSE syllabus. Notes for all subjects are available on the internet whether it is Maths, biology, political science, Indian history, Hindi etc. Get the course and structure and variety of chemistry notes in pdf form; free pdf download is also available for many chemistry textbooks, along with the entire science book. Therefore, to find an answer to any query, go online!

Finding A Private Tutor
A private tutor will help you get through different topics of chemistry with effective methods. Clarity of understanding is the most sought after demand of any student. A private tutor will help you achieve just that. You can also find chemistry teachers near you with zero application fee via Superprof, an online platform to search teachers.
Taking chemistry as your major from any university will not limit your career in any way. Chemistry aspirants can apply for government jobs, do different computer-based works, use your chemical knowledge in Engineering services, apply for UPSC or civil service exam or any other central government exam.
For the time being, wishing you all the best for your success at the SSC level. Especially in chemistry!
Résumer avec l'IA :