On reaching the SSC chemistry level, you will be required to learn and relearn a lot of concepts such as the atomic mass, atomic structure, the periodic table, biochemistry and so on. The task may look laborious but with the proper application of the useful method, it can be done quite easily. Chemistry is easier to understand when divided into multiple essential topics. For this reason, this article primarily focuses on the topic of chemical changes as part of the SSC chemistry syllabus.
The chemistry lessons and courses, offered by senior secondary boards, give special emphasis on the topic of chemical change. How well one does in the senior secondary level depends a lot on how well acquainted and familiar is he with the concepts of chemistry. For chemistry is a subject which has to be understood, first and foremost, on a fundamental level. If a student has managed to help himself with a good base in chemistry, it is very likely that he will perform excellently in the senior secondary level.
Chemistry may appear to be a subject which is highly complex but to understand it better what is required is to understand it on an elementary level. To be clear about the concepts such as what are atoms, molecules, protons, neutron, acids and bases, states of matter; what is electronic configuration, chemical bonding, covalent bonds, inorganic or organic compounds etc. is essential to go to the next level of chemistry. Apparently, chemistry is just like Maths: each and every step is necessary to progress to the next.
The topic of chemical changes is one such area which needs to be cleared in all its aspects. If you are planning to have an undergraduate research work in chemistry, this topic can not be taken lightly at all.
So without further ado, let us first learn about chemical changes in the context of general chemistry.

Defining Chemical Change
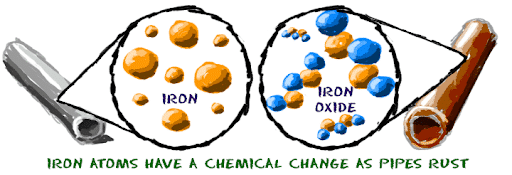
A chemical change is generally an irreversible chemical reaction where atoms of one or more substances are rearranged, resulting in the change in their chemical properties or chemical structure. The outcome of such chemical change in the substances results in the creation of one or more new substances. Unlike physical changes, chemical changes are irreversible in most cases. In those few which are reversible, further chemical changes are required to reverse the process.
An example of a chemical change would include burning of a candle into the wax, souring of milk, rusting of iron, etc.
Before we move on to the different categories of chemical changes, let us clarify certain things. The level of SSC chemistry depends on the premise of basic chemistry only. So do not be sceptical about relearning from the old class notes you have from previous year classes.
Now, let us move on to understanding a bit further about chemical changes.
Outcome Of Chemical Changes: Chemical Synthesis And Chemical Decomposition
Chemical synthesis is that chemical change in which chemical compounds are formed through simple ones. New products are created in chemical synthesis including the ones we need in our daily life. Chemical synthesis is mostly applied to organic molecules. Sodium (Na) and chlorine (Cl) combining to produce sodium chloride (NaCl) would be an example of chemical synthesis.
Discover more about some of the best chemistry classes in Coimbatore here.
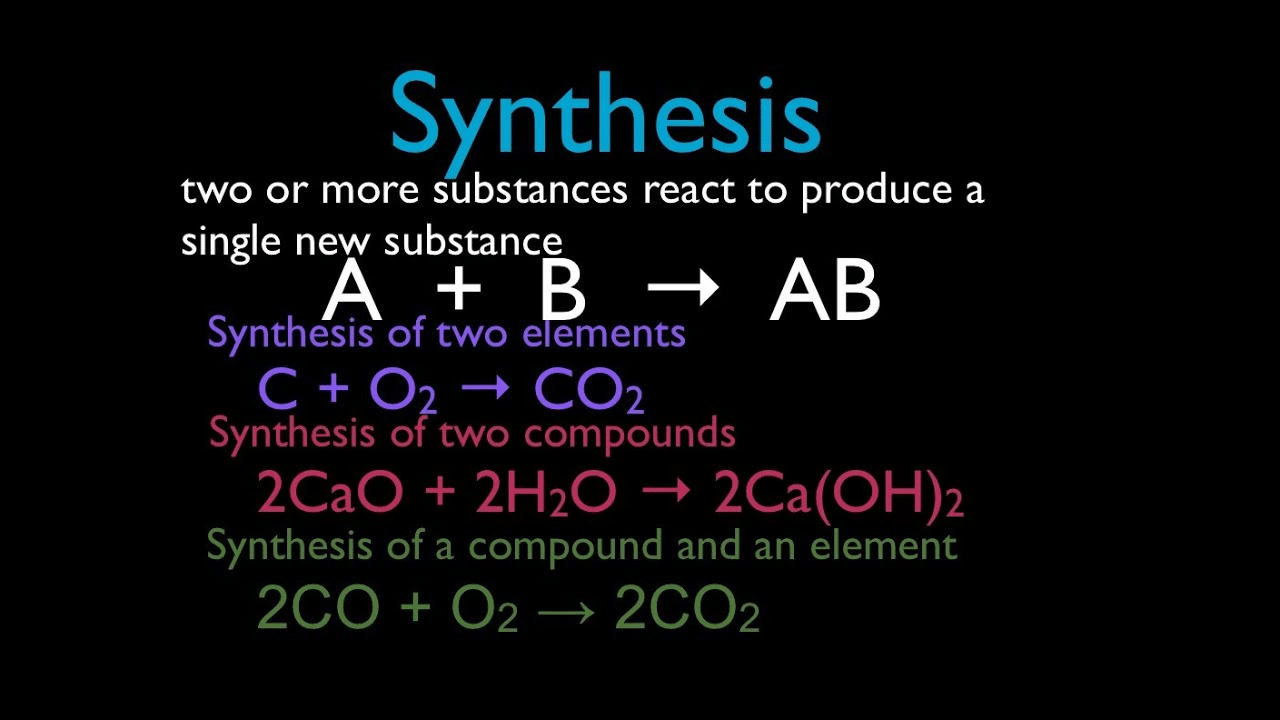
Chemical decomposition is the process in which a reactant or a single entity breaks down into two or more products. Electrolysis of water to hydrogen and oxygen is an example of chemical decomposition. Chemical decomposition is used in several analytical techniques such as mass spectrometry.
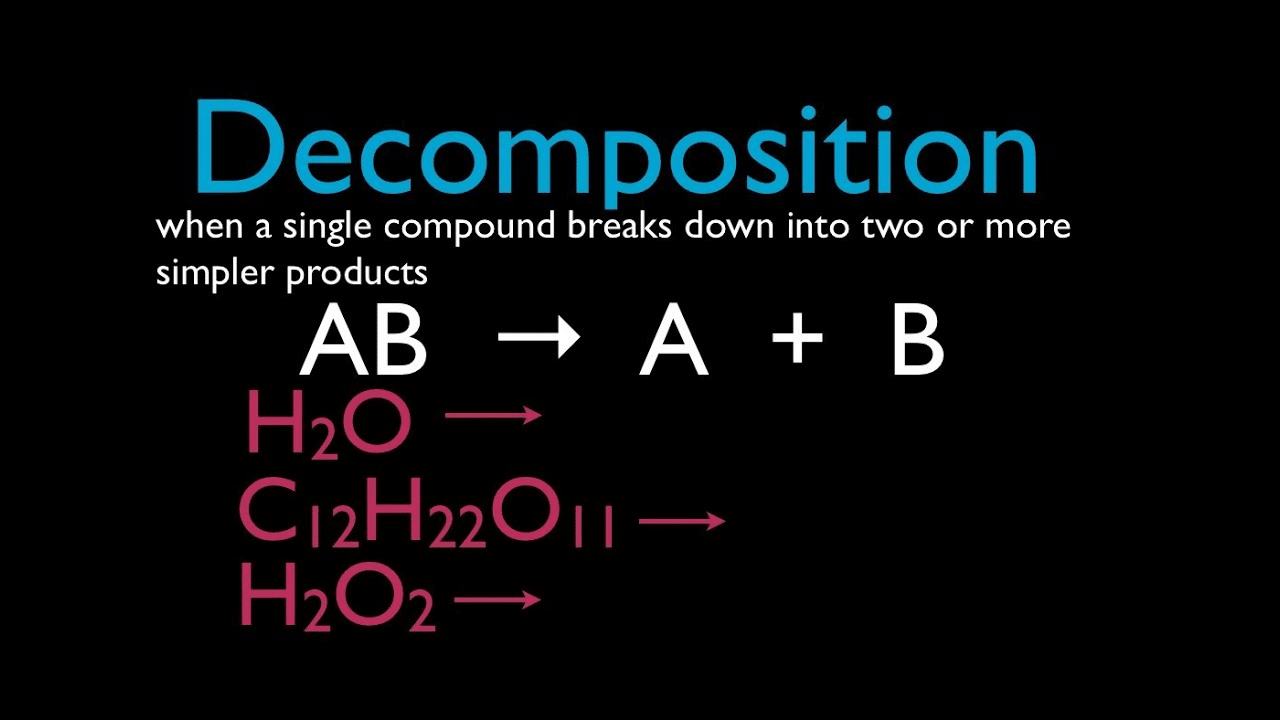
Both chemical synthesis and chemical decomposition are the alternative outcomes of chemical changes.

Energy In Chemical Changes: Exothermic And Endothermic Reaction
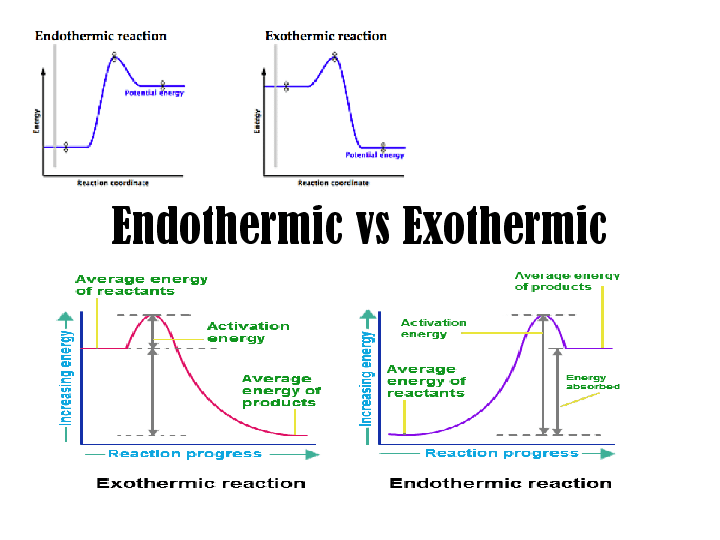
All chemical reactions involve energy. The energy is released/absorbed in the form of heat or light. The chemical reaction in which energy is released in the form of either light or heat is known as an exothermic reaction. The word ‘exo’ is derived from Greek and is literally translated to mean ‘out’, while ‘thermic’ implies heat. Therefore, any reaction in which heat is released, irrespective of the light being released or not, is known as an exothermic reaction.
The opposite of exothermic reaction is an endothermic reaction. In the endothermic reaction, energy is absorbed, generally in the form of heat, rather than released. This can be understood from the etymology of the word ‘endo’ derived from the Greek word ‘endon’ meaning ‘within.’ Photosynthesis or cracking of alkane would be an example of an exothermic reaction.
Find various chemistry classes on Superprof.
Types Of Chemical Changes
Now that we have understood the definition of a chemical change, in what ways it comes about and how energy is released in a chemical change, it is time to know about the type of chemical changes. Chemical changes are generally of three types: organic chemical changes, inorganic chemical changes and biochemical changes. Let us take a brief look at each one of them.
Organic Chemical Change
Organic chemistry tries to probe the structure, properties and reactions of different organic compounds, containing carbon atom in a covalent bond.
An example of organic change would be the burning of natural gas like methane. Methane reacting with oxygen in the atmosphere produces carbon dioxide and water.
Inorganic Chemical Change
An inorganic chemical change is the one where the reaction of different compounds and elements do not involve carbon atoms.
The lighting of the firework is an example of inorganic chemical change. A firework consists of metal nitrates which are burning compounds. When the firework is put to light, combustion takes place, which leads to the creation of new substances with the release of light and heat.
Biochemical Change
Biochemistry studies the chemical processes occurring within all living organisms. In biochemistry, most chemical reactions are controlled by enzymes and limited by hormones.
Photosynthesis is one of the most common examples of a biochemical change. In photosynthesis, plants convert light energy from the sun into chemical energy to produce food. Green plants convert carbon dioxide and water into oxygen and sugar.
Discover other chemistry online classes on Superprof.
Some Helpful Tips For Chemistry Preparation
Familiarity with the different chemistry topics will not come overnight. You will have to strive from your end as much as possible. Familiarity with SSC chemistry syllabus is the first step towards your learning. For instance, if you are from CBSE board, try checking out your Class 11 or 12 chemistry syllabus online. Based on the syllabus, you can focus on the particular areas step by step. On top of this, we have shortlisted some of the effective techniques which, if applied, could be really helpful to you.
Let us check out what they are-
Smartly Preparing The Chemistry Notes
Chemistry preparation is a thing which despite investing hours will not be as effective as a smart planned-way of preparing would be. This is because of the fact that the subject is theoretical on the surface only with an underlying set of rules, laws, and logical formulae. Also, it involves a lot of mathematics and also physic principles. So for you, the step would be to deal with the subject accordingly:
- Learn the atomic theory, valency, electron configuration, number of protons in different elements and other essential things which without memorizing you cannot use to solve chemical equations.
- Practice test questions from past papers to better understand the type of questions that may come in the chemistry exam.
- Be conceptually clear about branches such as organic and inorganic chemistry or physical chemistry and biochemistry and what makes them differ from one another.
- Do not miss out on chemistry lab classes.
- Prepare to keep in mind the career options you expect to have from chemistry. If in doubt, check out what chemistry majors offers as a career.
- Opt for a coaching centre or check out various online chemistry courses being offered by various institutes. They offer effective and efficient methods for quick revision of important topics in accordance with your syllabus.
Private Tutors And The Role Of Superprof
A private tutor can assess your strong and weak point about a subject - in this case, chemistry. Say, you are good in molecular formula, intermolecular forces and isotopes, the private tutor, then, will not lay extra emphasis on these topics but if you are weak in the concept of ionization or inorganic chemistry, he will spend more time in dealing with these topics only. This will help you overcome your weak points and lay the ground for a strong base in chemistry.
Finding a private tutor, that too for a particular subject like chemistry may be a bit time-consuming. Keeping this aspect in mind, online platforms like Superprof have come up as boon. At Superprof, one can not only find a private tutor for any subject but can also access the credibility of the teachers through their qualification and the reviews they got so far. One extra advantage that would come from using Superprof is that one can get the teacher from their own locality itself. All one needs is to enter their pin code and there you are!
We organised the topic of chemical changes categorically with the hope that this will make it easier for you to get through the basic concept of it. If it worked, do the same for the rest of the topics as well. Success will be assured!
Summarise with AI:















