For students who are aspiring to pursue the science stream, understanding of Chemistry becomes indispensable and essential. Until class 10, students are only introduced to a bare minimum knowledge of chemistry. The syllabus does not even scratch the surface of the subject. That is why we see some students who have performed well in class 10, struggle to barely pass in the senior secondary. It is only once they step in the senior secondary do they have to deal with the actual business of the subject. SSC chemistry is the beginning of fathoming the depths of chemistry.
The chemistry syllabus at the SSC level covers a number of topics. These include the structure of atoms, classification of elements and periodicity of properties, molecular structure, chemical bonding, chemical thermodynamics, equilibrium, states of matter redox reactions and much more. Any student wishing to attain a distinguished college for further studies in science must not neglect chemistry. It forms the basis of several other sub-disciplines that fall in, be it pure or applied chemistry. Besides, it requires much more than mere general knowledge about chemistry to crack any prestigious entrance.
Since these exams have no fixed or given syllabus, all ideal SSC chemistry courses cover all elementary focus points of chemistry and try to inculcate in the students a basic strong foundation. By the end of his/her school, a student is expected to know about organic and inorganic chemistry, chemical equations, electron configuration, kinetic molecular theory and topics alike.
Thus, studying seriously at the SSC level is most important if you are willing to make a career in the field of science.
In this article, we shall look at probably the most elementary yet most substantial topics in chemistry: atomic structure and the periodic table.
Discover more about some of the best chemistry classes in Varanasi here.
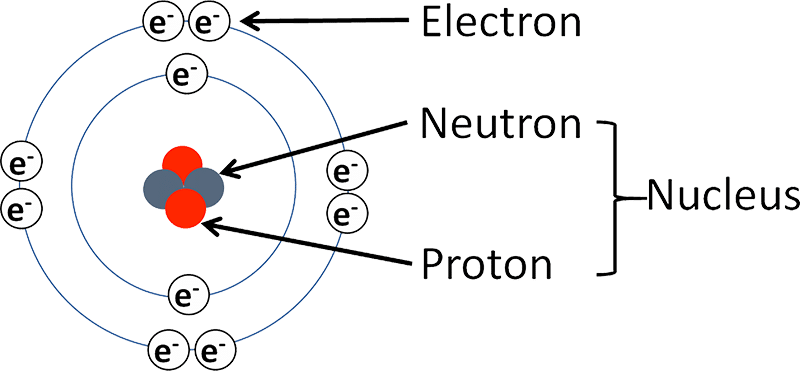

The Atomic Structure
Let’s not jump into complicated definitions and explanations of an atomic structure. We will begin with a simple Layman’s definition of it. Atomic structure is the entire configuration of an atom. We know atoms are the smallest constituent of matter but an atom in itself is made up of electrons, protons and neutrons which are even smaller than an atom. Imagine!
These protons and neutrons make up the nucleus in the atomic structure. Electrons are bound to the nucleus. The nucleus is positively charged owing to the presence of protons and the electrons are negatively charged.
Let us now go over the components of atom one by one-
Proton
A proton is marked with the symbol ‘p’. It is a subatomic particle which has a positive electric charge. It makes up the nucleus of an atom singularly or in more numbers. The number of protons (along with electrons) present in the nucleus of an element renders the element its basic characteristic. Each element has a different number of protons which makes up its unique atomic number.
Element A is different in properties from element B simply because of the varied number of protons that their atoms constitute of. The chemical reaction for each element would also vary accordingly.
Neutron
Just like the proton, a neutron is also a constituent of the nucleus. It has no net electrostatic charge and thus is often called neutral. Its mass does not vary much from that of a proton. With the exception of Hydrogen, neutrons are present in almost all atomic nuclei.
Protons and neutrons together are called nucleons. The subtotal of nucleons makes up the atomic mass number.
Electron
An Electron is a subatomic particle which is negatively charged. The electron has no known substructure or components and thus are known as elementary particles. Electrons just like every other elementary particle have properties similar to the wave-particle duality.
Electricity, magnetism, thermal conductivity etc. are influenced largely by electrons.
Now that we are familiar with the components individually and their properties, we shall delve a little deeper into the atomic structure. The electrons, being negatively charged are attracted to the positively charged protons by the electromagnetic force. On the other hand, it is the nuclear force that binds together the protons and neutrons in the nucleus.
Just like the number of protons determines the chemical nature of an atom, the neutrons determine the isotopes of an element. Besides all these, there is still much to learn about this subatomic particle.
Chemistry is the discipline which deals with investigating the properties of atoms like its ability to associate and dissociate. Arrangement of several atoms further makes up the molecular structure. Sound knowledge in nuclear chemistry is expected from people who wish to pursue research in atomic theory.
The Periodic Table
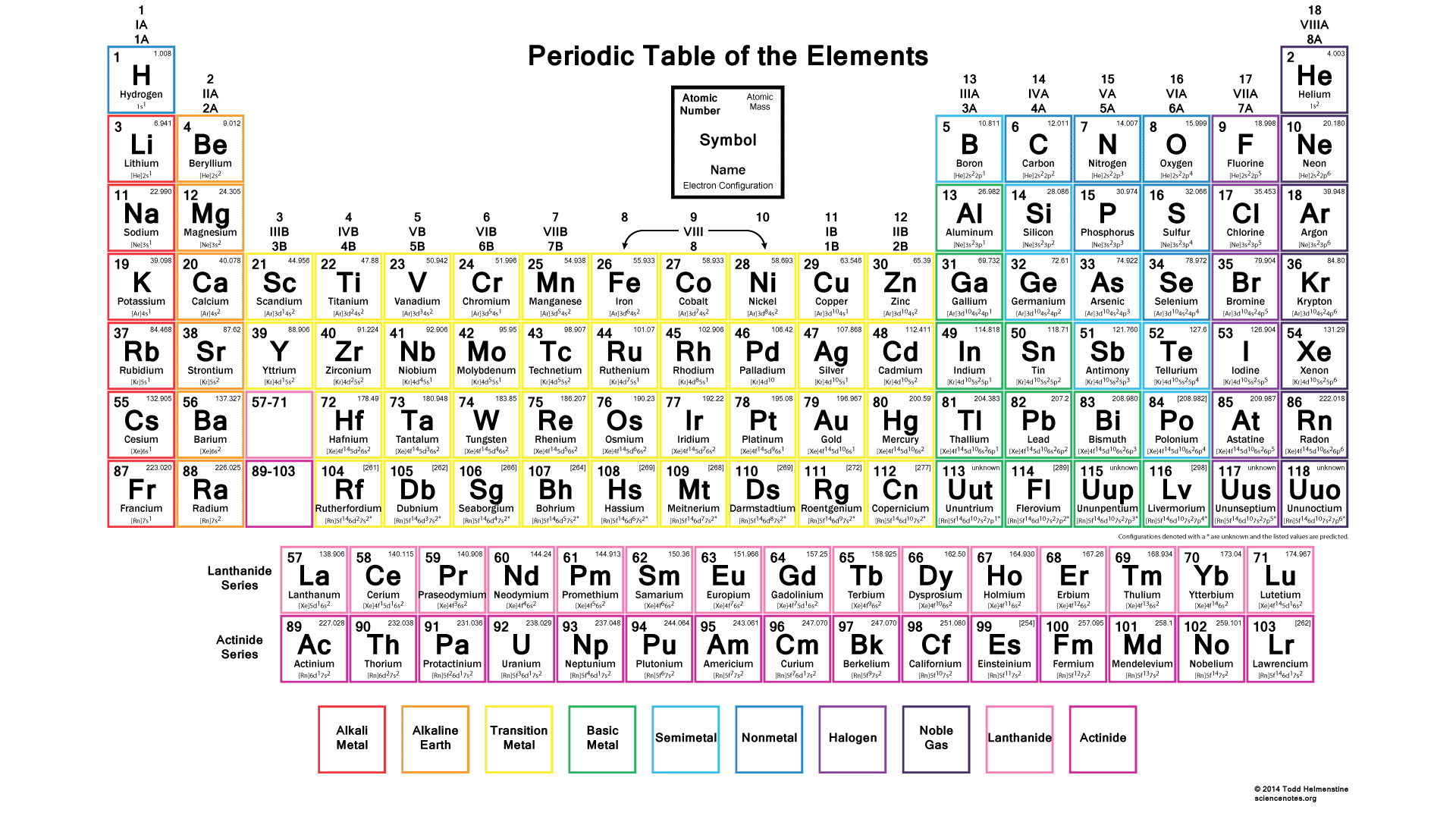
The periodic table of elements is a representation of chemical elements in a tabular form. These chemical elements are arranged by chemical properties, atomic number and electron configuration. The periodic table was formulated by Dmitri Ivanovich Mendeleev who was a professor of chemistry at Saint Petersburg State University. The table shows specific patterns that are the properties of those chemical elements. These patterns are called periodic trends. The table has seven rows which are called periods. The arrangement has metals on the left and nonmetals on the right.
The elements are further divided into groups. For example, group 18 contains noble gases. The primary 94 elements are all naturally occurring elements. Elements 95 to 118 are artificial synthesized elements. Which means they have been amalgamated in laboratories or nuclear reactors by chemical reactions.
Determining the valency of an element is important while studying the periodic table. Valency is a measure of combining the power of an element while forming a chemical compound or molecule.
Mere familiarity with the periodic table is not enough for students at the SSC level. They are expected to know the table like the back of their hands.
Click to join a chemistry class here.

Now that we are acquainted with basic chemistry, let us focus on how to secure the best possible marks in the SSC examination.
Exploit Your Best Options
In order to be a distinguished student of chemistry, one cannot just rely on the chemistry classes conducted by the school. The students should independently move forward with chemistry courses and practice and revise at their respective convenience. Let us quickly go through the tips, if followed, can help you pass your exams with flying colours.
Focus On Your Own Preparation Besides School
Try to be regular at classes and clear your doubts with teachers as much as possible. Apart from that, keep going through past papers and test questions. Also, solve practice test papers of chemistry questions. If interested, you may also browse through several chemistry journals. You may appear for competitions like the Olympiad. You will be required to answer multiple-choice questions, which will boost your confidence and compel you to do even better. Widen your knowledge of the history of chemistry.
These will not just help you pass your SSC examination but will also make your foundation strong which in turn will help you later as a graduate student or in your chemistry research.
Seek Help Of A Private Tutor
Chemistry faculty of all schools are not alike. Some may have teachers who are impeccably good and some may not. It is unfair for students to suffer the loss of quality in imparting education.

Everyone has their own inclinations and aptitude. One student may be very perceptive of organic chemistry but comparatively bland about atoms and its structure. One must not expect the school to cater to the individual needs of every student. In these circumstances, one can always seek assistance from a private tutor.
An online search for private tutors is the newest thing on the internet and it has very positive feedback. Superprof is one such platform that can provide you with eligible tutors who live close by. It is a hassle-free business as you need not go through a tedious appointment. All the interaction is done through the website itself.
Also, you need not hire a teacher without any background check. Superprof provides you with the applicant's academic degrees and achievements. To top that, you may also go through the reviews he/she has acquired. All these crucial factors make Superprof an extremely reliable platform for parents to entrust their children’s career with.
The platform harbours a ton of talented tutors who can guide students to realize their aspirations and dreams.
Discover other online chemistry classes on Superprof.
Tips For The Examination
Prior to this section, we have been talking extensively about the preparation. Let’s now talk about the examination itself. Here are a few tips which will surely help you score well:
- Cover all the chapters in your SSC chemistry syllabus. Do not rely on your luck. Chemistry is an intricate and conceptual subject hence overlooking portions may lead to a disbalance in your entire understanding faculty.
- Try to answer all the questions. Solve question papers at home to master time management at the examination hall.
- Make a notebook and revise all the topics the day before the examination. Do not panic.
- Do not try to memorize chemistry. It is supposed to be understood.
- Do not hesitate to seek help from your teachers.
- Try to keep 90 your target score and work hard accordingly. Even if you score in the 80s, it is pretty good with room for improvement.
- Lastly, do not get demotivated at any point in time. “Read, revise and recapitulate” should be your motto.
Chemistry, if the basics of it are understood, is not just a relatively interesting subject but even a great career option. Notably, new initiatives are being taken in India to make the subject more interesting and fun. So, all you will be required is to opt with a firm goal in mind and prepare accordingly. The choice is all yours!
Summarise with AI:















