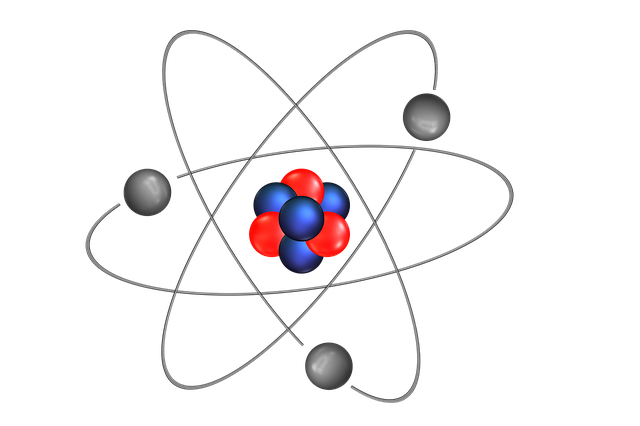
As we know all the matter around us is made up of the atoms. These atoms are present in solid state, liquid state, and gaseous state. When studying solid state physics or the other fields, the atom can be further divided into smaller classifications. Most important for a class 10 physics student is learning the structure of an atom. An atom consists of protons that are positively charged, electron that is negatively charged, and neutrons that have a neutral state. Protons and neutrons are present at the center of the atom and the electrons are floating around the nucleus.
There have been several scientists who have given various theories depicting the structure of an atom. The study of an atom comes under particle physics and in it, a particle related study is done. This is also known as matter physics. The atomic structure has now been a part of quantum physics and quantum mechanics leading to the invention of further subatomic particles. The study of physical science is quite interesting and in this article, we will cover the class 10 physics concepts about the atomic structure. Check the syllabus of class 10 physics.

Atomic Structure: Definition

An element’s atomic structure is defined as the constitution of protons, neutrons, and electrons and their arrangement in an atom. The nucleus of the atom is made up of protons and neutrons that are surrounded by free-floating electrons. Atoms that are neutral have the same number of protons and neutrons in them.
The atoms though lose or gain the electrons in order to become more stable. The resulting entity is then called an ion. Various elements that are found on earth have different atomic structures due to the presence of different numbers of electron and protons in them. This is why every element is considered unique.
Various Atomic Models
During the 18th and 19th century, there were numerous scientists that were fully involved in trying to explain the structure of an atom. Many scientists tried and failed, but few were there whose atomic models were found to be logical. Amongst them, J.J. Thomson, John Dalton, Niels Bohr, and Ernest Rutherford, were the ones whose contributions were found to be the most accurate. We will discuss their models of the atomic structure further below in the article.
Atomic Theory of Dalton
John Dalton, an English scientist first suggested that all the matter of the universe is made up of atoms that are indestructible. Further, he explained that all the atoms of similar elements are the same and the atoms of two different elements differ from each other. The mass and size of the different elements are different. In the case of chemical reactions, rearrangement of atoms occurs due to which a new product or element is formed.
Below given pointers are the main understandings of his theory:
- Atom constitutes of all the matter.
- Atoms can not be divided further.
- Only one type of atoms is there in specific elements.
- Every atom has its own mass.
- At the time of a chemical reaction, there is a rearrangement in the structure of an atom.
- Atoms can neither be created nor destroyed but can be transformed from one form to another.
Negatives of Dalton’s Theory
- His theory could not explain the formation of isotopes.
- The structure was not explained thoroughly.
- Later it was found that an atom could be divided further away.
Atomic Model of JJ Thomson
In the early 1900s, JJ Thomson an English scientist proposed his atomic structure to the world. He was also awarded the Nobel Prize later for his discovery of electrons based on his experiment of cathode ray tube.
JJ Thomson Experiment
A glass tube with two openings is taken, one is for the pump (vacuum) and the other one is for the inlet (it helps in inserting the gas). The vacuum pump maintains a level of vacuum inside the tube. Electrodes are fitted inside the tube and are connected with a high source power.
After the experiment was done, JJ was able to prove that there is an emission of negatively charged particles when the experiment is done. Those negatively charged particles were known as the electrons.
They had some mass and energy of their own was shown in the experiment. When the electric field and magnetic field were applied to the cathode way tube charge to mass ratio of electrons came out to be 17588 × 1011 e/bg. The experiment was done in a physics laboratory under his JJ Thomson supervision.
Based on the experiment, JJ concluded that elements have a positive body in them around which negatively charged particles are embedded around. He stated that both negative and positive charged particle has a similar magnitude inside an element. His model is also known as the plum pudding model.
Although Thomson was able to discover the electrons, his model could not explain how was an atom stable. Also, when later subatomic particle was discovered they could not get fit into his model.
Find out about class 12 Physics curriculum.
Atomic Model of Rutherford
In class 10 physics, you will also study Rutherford’s atomic model based on the principles of physics. Rutherford was a student of JJ Thomson and he updated the model of Thomson by adding a nucleus to the model. He did an alpha ray scattering experiment that showed the presence of a nucleus in an atom.
As per the experiment results, it was suggested that most of the mass and charge of the atom is there in the nucleus. His theory suggested that:
- The mass and charge of an atom are concentrated at the center known as the nucleus.
- The structure of an atom is spherical.
- Electrons are continuously revolving around the nucleus of an atom like the planets are revolving around the sun.
However, his theory could not explain that if the electrons are continuously moving around the charged particles, then they would lose energy. On losing energy they will fall back to the nucleus and the atom can not sustain itself. Also, in reality, the spectrum that was noticed was the line spectrum but his theory suggested it to be a continuous spectrum.
Find a great physics tutor here on Superprof.
Atomic Model of Bohr
In the year 1915, Neil Bohr put forth his atomic model that explained the structure on an atom. To date, this atomic structure is considered the most genuine and right adaptation of the structure of an atom.
His theory suggested that:
- All the electrons move inside the atom in a stationary orbit.
- There are different energy levels or shells in which the electrons rotate.
- Electrons constantly jump from one level to another on the loss or gain of energy.
- When an electron gains energy, it jumps to a higher level, whereas when it loses energy, it goes to the lower level.
The atomic model of Bohr could not explain the behavior of an atom during the presence of the electric or magnetic field. It also helps to understand the particle model in class 10 physics.
Learning Basic Physics Concepts

In class 10 physics, the need to learn the concepts and make them stronger is very important. This is so because when we go forward to study high school physics, topics like thermodynamics, magnetism, kinematics, special relativity, differential equations, quantum field, etc. comes into the picture. You can learn the concepts of electricity here. If the basic concepts of laws of motion and other physics concepts are not properly known to a student, they have to struggle in higher classes. So it is very important for a student to thoroughly understand the concept of physics in their class 10 physics. Here are a few tips for students to score good marks in the exam.
Mastering Physics Online Through Superprof
Physics is an important subject for every one of us as it is related so much to our daily lives. So, for a student who is currently studying in their 10th standard, it is of high importance to learn all the concepts of physics. For it, a student can take various routes to make his understanding better. Learning physics online is a great option for students. Many times the student is not able to understand the concepts properly and ignore it. Later it becomes huge trouble for them. Study on Superprof the various concepts of forces.
So with the help of Superprof, one can opt for an online teacher who can help the student to clarify his doubts, learn the concepts, and get good marks in the exam. Superprof has thousands of teachers online whose profiles and testimonials from the students can be checked. Superprof helps to pick the best teacher for a student in their own locality or area. Find the concepts of different waves, sources of energy and concepts of electromagnetism on Superprof.
Also, the first session on Superprof remains free so it becomes easier to check the way of teaching of a teacher for a student. A student can check if he is able to understand the methodology of teaching and therefore can make an efficient decision for himself. So in order to get the best grades in class 10 physics, Superprof is undoubtedly the best way.
See the perfect Physics class for your kids.
Summarise with AI: