Chemistry is a class you take in high school or college, where you figure out two plus two is 10, or something. - Dennis Rodman
Chemistry is packed with complex words and phrases that require understanding to grasp the subject as a whole. It can be a little difficult to understand and learn all the words in a short span of time but once you get your grips on the glossary, everything else becomes much more lucid. It makes wrapping your head around the concepts discussed in your chemistry classes easy.
However, you need not be overwhelmed with the scientific vocabulary and proceed little at a time starting with basic terms. Once you start feeling comfortable with the basic ones, learning more complex terms becomes easier. Unless you pay attention to the vocabulary, no amount of chemistry tuition can help you crack the entrance exam you are aiming at.
It is imperative that you know these definitions. Only then can you comprehend the confusing world of reactions, substances, experiments and laboratory work called chemistry. Even if you know the terms well, you should polish your skills by occasionally brushing through them. This will help keep concepts fresh in your mind.
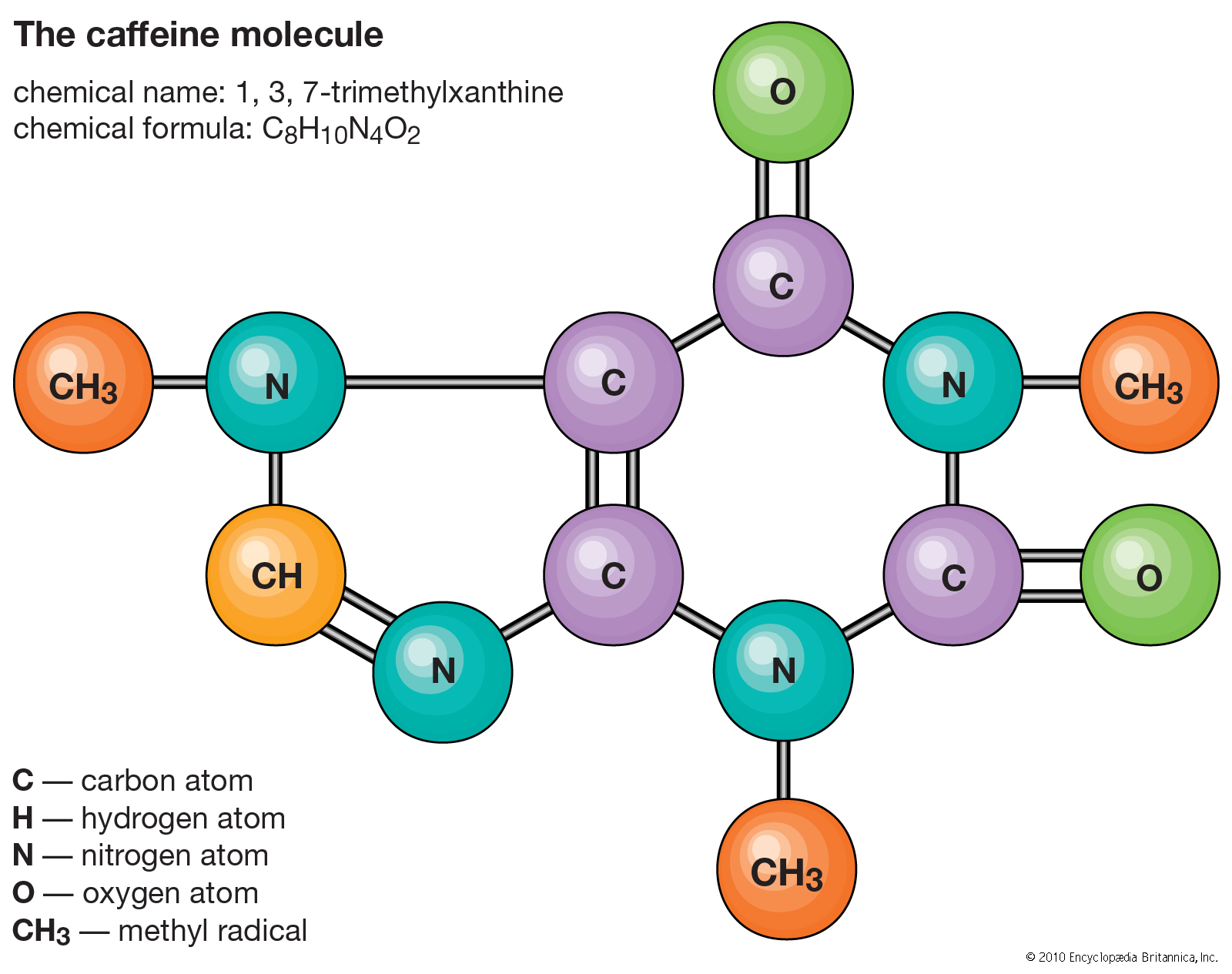

Basic Chemistry Terms to Learn from Entrance Exams
Students who are preparing for undergraduate level or university science courses need to ace their laboratory skills, strengthen their research on organic chemistry and physical chemistry to fulfil the entry requirements of top colleges.
But before you study and can fully grasp either organic chemistry, physical chemistry, or other segments, you must be familiar with some very basic English language terms pertaining to the subject. Here is a list of basic chemistry terms to learn from entrance exams:
Molecule
A molecule is the number of atoms that are bonded together in a chemical bond and are electrically neutral.
Acid
Acid is any compound that gives a pH level of less than 7.0 when dissolved in water or donates a hydrogen ion.
Mixture
A mixture is a material that we get after mixing two or more different substances. The substances in the mixture are mixed physically but not combined chemically.
Liquefaction Point
The liquefaction point is also known as the melting point. It is the temperature that changes the state of a particular substance from solid to liquid. The temperature also depends on pressure.
Filtration
Filtration is the process that separates large particles from smaller ones. It could be either a physical, biological or chemical operation. It is done by passing the particular mixture through a well-constructed and complex lattice structure which filters out the small size while retaining the larger ones.
Distillation
Distillation is the process that is used to separate the component substances of a liquid mixture. It is done by exploiting differences in the volatility of the components present in the mixture through selective boiling followed by condensation. A still is used to conduct this process.
Find chemistry classes here on Superprof.
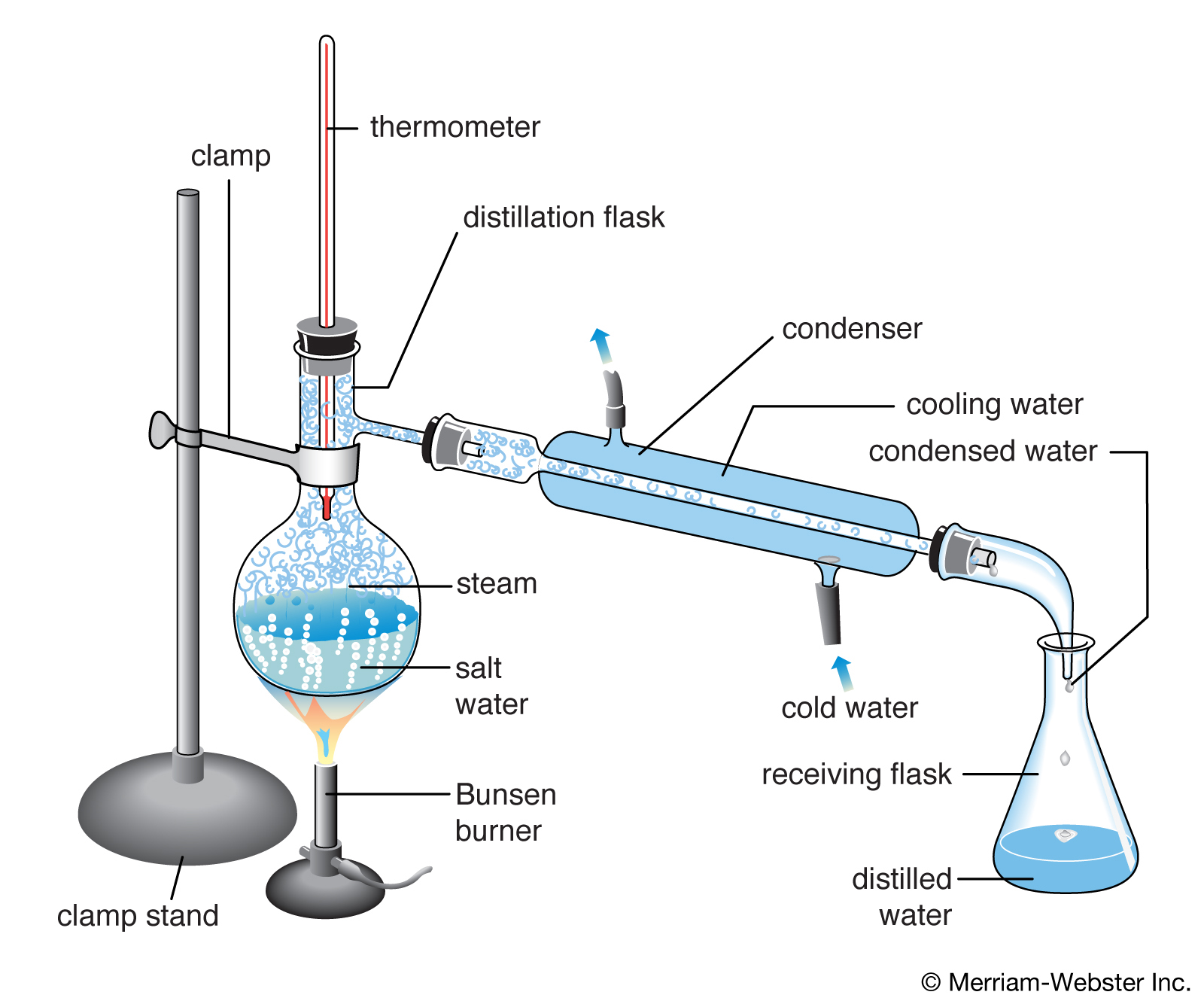
Compound
The compound is a substance that arises from chemically bonded elements.
Advantages of Having a Home Tutor
Any academic year for chemistry students can be overwhelming if they do not have the right kind of help. Whether you are pursuing Chemistry BSc Hons or studying for your 10 plus course, you can go a long way with some additional help besides your school or college.
Chemistry courses include a lot of things and it is often not possible for the school to give individual attention to every student. However, when you enter the world of entrance exams, you cannot take a chance or make a compromise. You have millions of students competing against you, even international students.
Thus, it is imperative that you hire a home tutor who can help you out. If you are still not sure, here are some of the reasons that will put adequate reason into this argument.
Undivided Attention
Schools and other institutions abound in students which makes it difficult for the teacher to focus on a single student. With a home tutor, you can have his undivided attention. This means no matter how many times you get stuck in a particular segment or chapter, he is there to help you out.
A home tutor’s teaching style varies from one student to the other. This individualised approach is impossible in a school. With tuition, you get to target your weak points and strengthen them.
Study at Your Pace
Not every student in a class has the same pace of comprehending. Some might be slower than others. When taught in a large group, such students suffer from disadvantages. But when you study privately, your tutor knows your pace and proceeds accordingly.
Besides, school and college teachers rush to complete their syllabus which might negatively affect the quality of teaching. But with a home tutor, you need not worry. He will take time and move over to the next chapter only when your ideas about the previous one are crystal clear.
Bonding
A strong teacher-student bond can skyrocket the latter’s interest in the subject. Students often do badly in a subject not because they are not smart but because they fail to find that subject interesting enough. Such teachers also stand out as idols and role models which further the student to do better in their fields.
Doubts Clearing Sessions
Clearing doubts about each chapter as you move forward is very important. All the chapters are interrelated and you cannot have a good overall understanding unless you have your concepts clear. A private tutor can clear your doubts not once but as many times as you want.
You can also ask for extra classes in times of exams. Besides that, you can seek help for your research project from him. With expert insight, it could help you spike up your grades significantly for better career possibilities.
Convenience
Managing all the subjects can put a strain on your mind if you do not have a sorted timetable. You have no control over the classes your educational institution arranges but in the case of private tuition, you have the convenience of setting the time and date according to your timetable.
You and your tutor can discuss and come up with a time that will suit both of you. You need not even skip class the day you are unwell or out of the station. You can simply reschedule it for some other day. This way you do not miss out on any important class.
Practice and Revision
Practice and revision are integral parts of science. You must keep brushing up your formula and revise the previous chapters taught. If you get stuck, your teacher will help you out.
Find chemistry classes here on Superprof.
Why Chemistry Online Classes are the Best Option?
Chemistry online classes work best for both the teacher and the student simply because it saves the time one would have to invest in travelling. Online classes have their own advantages. It saves time and money which effectively reduces your tuition fees to some extent. It makes learning easy and fun. The benefits of online chemistry classes are:
- Flexibility: Online classes are more flexible than offline classes, which comes with a rigid schedule.
- Comfort: Online classes are convenient in terms of tavelling. Also, you do not have ant boundary.
- Time-saving and cost-effective: Travelling is time-consuming, which is not an issue in the online class. Also, online learning is more cost-effective since you get better attention.
With the online class, you can also have less pressure of studies and no worries in terms of any physical or external factors.
Find chemistry classes here on Superprof.

How to Find Chemistry Classes Near Me?
Students often look for tutors on google by typing ‘find chemistry classes near me’. While it can yield you results, not every suggestion will be an authentic and verified one. Several tutors put up their advertisements on the internet and their claims on achievements and degrees may not be genuine.
Since there is no way to know, you might end up appointing the wrong tutor. Therefore, look for a platform that ensures you can verify the claims that these teachers make.
Superprof is one such platform that works with the aim of delivering the best teachers to ambitious students. They have a long list of teachers ranging from all over the country. Since the classes conducted these days are mostly online, you need not restrict your search to your geographic location. This way you can take classes from the best teacher.
The website lets you check the background of each teacher listed. You can see through their profile and verify their degrees, accolades and achievements. You do not have to trust ads or word of mouth and blindly hire a teacher. This gives you the chance of comparing teachers and select the one that you think will work the best for you.
Besides that, you can also see the ratings and reviews that the previous students have left on their profiles. This gives a clear idea about how dedicated and sincere the teacher is. They can be the torch bearers of the next generation and help the students achieve great heights.
Summarise with AI:















This is too helpful.
I am really impresded with it.
bookmarked!!, I like your site!
There’s definitely a lot to know about this issue. I really like all the points you made.